 This force is present whenever there is relative motion between the fluid surfaces. This resistance to motion in liquids is known as viscosity, and it can be thought of as internal friction similar to that created by motion on the surface of solids. 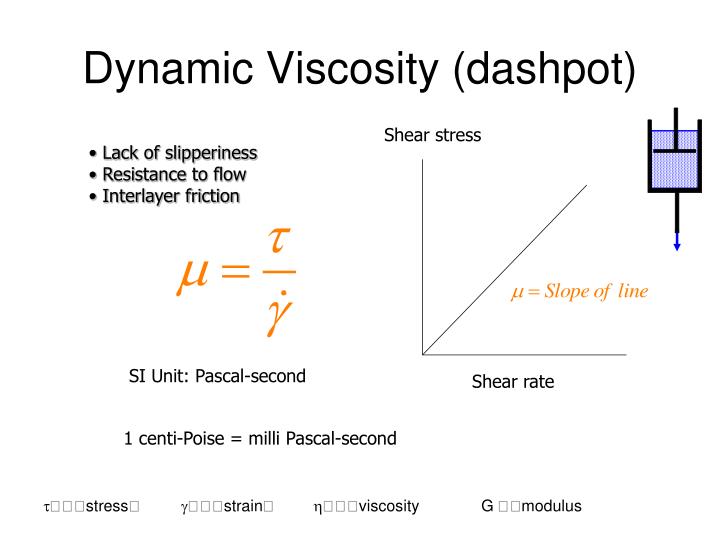
Because not all liquids are ideal liquids and because they exert some resistance to motion, the moving fluid has numerous characteristics that prohibit it from being an ideal liquid. Fluid in motion possesses a variety of qualities and contains a variety of energies. Internal friction is another name for this force, which acts between liquid layers. Viscous force is the name for the force caused by viscosity. The characteristic of liquids known as viscosity prevents relative motion between its many layers. The characteristic of liquids that stops them from spreading is their viscosity. This occurs as a result of the two fluids' differing viscosities. Honey would require more time to shape the bowl than water would. Let's say someone had two bowls, one of which is filled with water and the other with honey. Ideal or inviscid fluids are those with no viscosity. Superfluids are the only fluids that exhibit zero viscosity, or no resistance to shear stress in all other fluids, positive viscosity is required by the second law of thermodynamics. For instance, the rate of deformation does not affect the viscosity of a Newtonian fluid. In some circumstances, the dependence on some of these traits is minimal. In general, a fluid's state-including its temperature, pressure, and rate of deformation-determines its viscosity. This friction caused by a fluid moving against it is comparable to internal friction, which is comparable to friction caused by a solid moving against a surface. The majority of this liquid has some motion resistance and is not ideal. When the liquid layers are moving relative to one another, there is viscosity. The assessment of a fluid's resistance to a progressive deformation caused by tensile or shear stress is known as viscosity. It is possible to think of viscosity as the friction that occurs between fluid molecules. A molecule's kinetic energy increases with increasing temperature, which causes molecules to slide past one another between layers. A liquid's viscosity reduces as its temperature rises. Viscosity quantifies the obstruction to a liquid's flow that results from internal frictions between fluid layers. Viscosity is also present in gases, but it is less obvious in everyday situations. A fluid with low viscosity, on the other hand, flows smoothly because its molecular structure causes less friction when it is in motion. Strong intermolecular forces provide a lot of internal friction in a fluid with a high viscosity, which makes it difficult for layers to move past one another. The term "viscosity" can also be used to describe a fluid's thickness or resistance to being penetrated by foreign objects. More specifically, it measures flow resistance brought on by internal friction between fluid layers that occur when they pass one another during fluid flow. Oxygen dynamic and kinematic viscosity at atmospheric pressure and varying temperature:ĭynamic viscosity of oxygen at varying temperature and 1, 10, 50 and 100 bara (14.We refer to this resistance to motion that most fluids provide as "viscosity." When there is relative motion between the fluid's layers, viscosity develops. See also other properties of Oxygen at varying temperature and pressure: Density and specific weight and Specific heat (heat capacity), and Thermophysical properties at standard conditions,Īs well as dynamic and kinematic viscosity of air, ammonia, benzene, butane, carbon dioxide, ethane, ethanol, ethylene, methane, methanol, nitrogen, propane and water. 
Temperature Choose the actual unit of temperature: While the kinematic viscosity is given as cSt, m 2/s, and ft 2/s The output dynamic viscosity is given as Pa*s, N*s/m 2, cP, mPa*s, lb f*s/ft 2 and lb m/(ft*h), The calculator below can be used to estimate oxygen dynamic or kinematic viscosity at given temperatures and atmospheric pressure. Oxygen phase diagram Online Oxygen Viscosity Calculator Tabulated values and viscosity units conversion are given below the figures. Absolute or dynamic viscosity is used to calculate Reynold's Number to determine if a fluid flow is laminar, transient or turbulent. 
The viscosity of a fluid is a measure of its resistance to gradual deformation by shear stress or tensile stress.įor further definitions, go to Absolute (dynamic) and kinematic viscosity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
